Corneal Cryopreservation
Background
Corneal transplantation addresses numerous ophthalmological diseases, e.g., Fuchs dystrophy, keratoconus, infectious keratitis, and damage due to corneal trauma damage. Worldwide, over 200,000 corneal transplantation procedures (approximately 100,000 in USA) are being performed each year.
For over 90% of corneal transplants, maintaining the quality of the endothelium of donor tissues is critically important: the endothelium of corneas is the most posterior layer, comprising an approx. 5 μm thick monolayer of mitochondria-rich and non-regenerative cells. These cells are critical for regulating fluid and solute transport between the aqueous and corneal stromal compartments. Currently, hypothermia (2-8°C) is the commonly applied corneal storage method, but only allows for effective storage of the endothelium for 10 to 14 days, which results in numerous operational and clinical issues:
- The cold-chain management associated with the short bench life poses a heavy financial burden to eye banks for management and transportation;
- Eye banks work within the limits of their inventory to requesting surgeons and the quality of tissues is not always guaranteed;
- The short bench life only allows screening of general transmission diseases based on standard tests of donor blood. However, a wider spectrum and more in-depth tests on donor tissues, where rare but lethal viruses can reside, is always desired but impractical;
- Worldwide, there are over 10 million patients needing corneal transplantation. However, for many developing countries, due to either cultural or policy restrictions and technical limitations, domestic corneal donation or collection is minimal, and it is highly challenging for their patients to obtain qualified corneas from USA or other developed countries. Currently, 30% corneal tissues (over 60,000) donated in USA are transplanted overseas, but the quality of those tissues, after extended period of transportation and processing, requires significant improvement
- For medical or fundamental research (around 30,000 donor corneas used annually for these purposes in USA), quality-controlled tissue is either not available or unaffordable.
Currently, there are no commercial technologies on the worldwide stage that would allow corneal tissue to be cryopreserved for clinical use. While there are protocols by which corneal tissue can be frozen with extremely high concentration of glycerol, these tissues have no viable endothelium cells and are only mostly used as special surgical tools. Traditional cryopreservation technology not only requires liquid nitrogen facilities for storage and transportation, which is unacceptable to current stakeholders, but also suffers from endothelial cell loss of approximately 30% or more, while current clinical practice for transplantation requires over 90% viability of endothelial cells. As a result, even after decades of effort, cryopreservation of corneas to maintain viability of endothelium has failed to gain wide acceptance.
With the development of bioartificial corneas, esp. those composing an engineered endothelial cell layer on a biological or artificial tissue, which will potentially finally satisfy the demand for millions of corneal transplantations globally in the future, the current hypothermia treatment will then be found to be incapable of storing and transporting the large number of those bioartificial tissues in a potentially huge demand. That inefficiency will tremendously impede the future bioartificial corneal transplant practice and thus impact the life quality of millions of patients.
Innovation Based On IcyEye® Technology
Seeking FDA Clearance
Using CryoCrate’s IcyEye® kit (see Technology for more details regarding the working mechanism), human corneas can be efficiently cryopreserved in regular lab deep freezers (-70°C to -80°C) for long-term and safely transported in dry ice boxes (-78°C on the dry ice surface) for international shipping and processing. As the parts of the kit, the novel container design removes the growing ice front that damages endothelium mechanically, and the C80EZ®-CORNEA medium enables long-term cryopreservation of endothelium in freezers and minimizes ice crystal size inside the tissues. The kit is disposable and “good-for-one-time-use”, to prevent potential contamination or disease transmission due to misuse or re-use. After long term storage of human corneas at -80°C using IcyEye® kit, the standard endothelial cell count is approaching 100% of that prior to freezing, and tissue integrity and clarity is not impacted by the cryopreservation procedure. The C80EZ®-CORNEA medium is also compatible with existing hypothermia media (e.g. Optisol) that are currently used for cornea harvest and transportation by eye banks, and as such, the thawed corneas can be directly transferred to those media for further processing or analyses.
We believe the IcyEye® technology will solve the current practical and clinical issues related to corneal transplantation and pave the path to the future application of tissue engineering in this industry as well as other biomedical applications of bioartificial tissues.
CryoCrate is currently seeking FDA clearance for its adoption in the USA market.
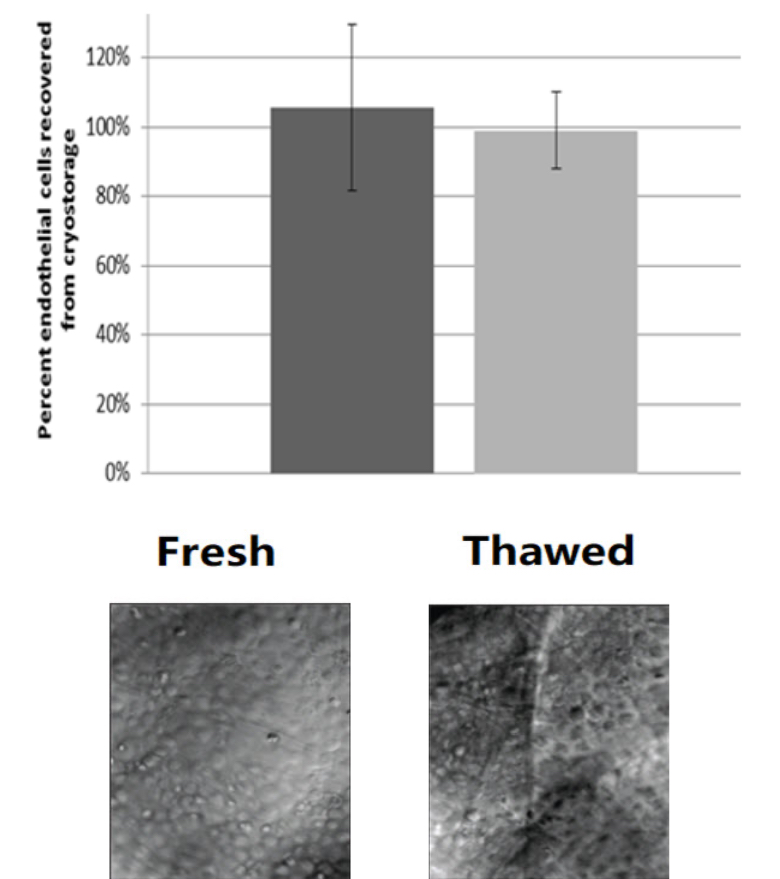
A comparison between the endothelium morphology and endothelial cell count between fresh tissues (Left) and thawed tissues from IcyEye® (Right).
